Crystalloluminescence
What is crystalloluminescence?
There are a lot of different types of luminescence, where light is emitted by some process, e.g., chemiluminescence where light is given out by a chemical reaction. Crystalloluminescence (XTL) is light given out during crystallization.
There are a lot of different types of luminescence, where light is emitted by some process, e.g., chemiluminescence where light is given out by a chemical reaction. Crystalloluminescence (XTL) is light given out during crystallization.
Why have I not heard of this before? Is it important?
Remarkably the earliest reports of XTL go back as far as 1787! In olden times chemists were much more likely to be working in dark conditions, and so phenomena involving emission of light may have been easier to see. Brandowski was the first to report XTL of NaCl in 1894. The importance of XTL is that each emission of light represents the birth of a new crystal, and therefore can tell us something about the dynamics of the nucleation process. The animation below illustrates the flashing emissions for NaCl.
Remarkably the earliest reports of XTL go back as far as 1787! In olden times chemists were much more likely to be working in dark conditions, and so phenomena involving emission of light may have been easier to see. Brandowski was the first to report XTL of NaCl in 1894. The importance of XTL is that each emission of light represents the birth of a new crystal, and therefore can tell us something about the dynamics of the nucleation process. The animation below illustrates the flashing emissions for NaCl.
How strong is the crystalloluminescence?
The luminescence is very weak. In order to see it you need to get your eyes well accustomed to the dark. The image below shows XTL of NaCl taken with 15 s exposure using a compact digital camera [Canon S90, f/2, ISO 3200]. To create a rapid crystallization, concentrated HCl (24 wt. %) is mixed rapidly with slightly undersaturated (98%) NaCl solution.
The luminescence is very weak. In order to see it you need to get your eyes well accustomed to the dark. The image below shows XTL of NaCl taken with 15 s exposure using a compact digital camera [Canon S90, f/2, ISO 3200]. To create a rapid crystallization, concentrated HCl (24 wt. %) is mixed rapidly with slightly undersaturated (98%) NaCl solution.
What colour is the emission from XTL of NaCl?
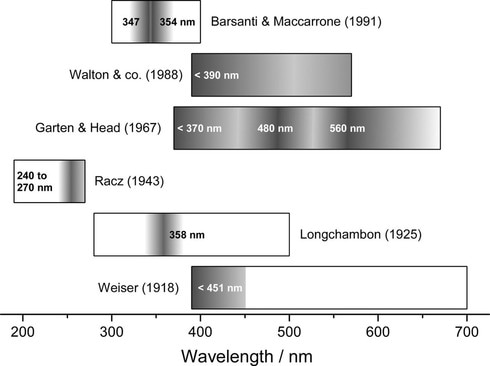
We were the first group to determine the complete XTL emission spectrum for NaCl [1]. Up to this time, researchers had given different accounts of what the colour was, although most agreed that it was blue or blue-green. The confusion is summarized in the image below.
We were the first group to determine the complete XTL emission spectrum for NaCl [1]. Up to this time, researchers had given different accounts of what the colour was, although most agreed that it was blue or blue-green. The confusion is summarized in the image below.
What is the mechanism for XTL from NaCl?
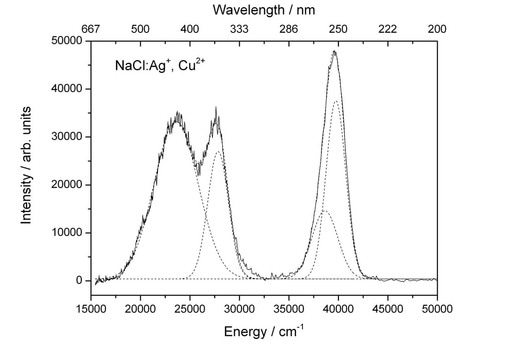
Remarkably enough it is not the NaCl that is emitting the light, it is impurity metal ions, such as Cu(I)/Cu(II) and Ag(I). This explains why the effect appeared to be slightly different colours in the past, as the effect would depend on the quality of the water supply! The spectrum below shows peaks in the emission. The solution was intentionally doped with ppm of Ag(I) and Cu(II) ions. The peaks can be attributed to the ion centres trapped in the crystal: at 252, 259 [Ag+] and 359 [Cu+] and 424 nm [(Ag+)2 dimers]. What this tells us is that these ions are trapped in their excited states during rapid transformation from disordered NaCl solute cluster to ordered NaCl crystal. For more details, see the further reading [1].
Remarkably enough it is not the NaCl that is emitting the light, it is impurity metal ions, such as Cu(I)/Cu(II) and Ag(I). This explains why the effect appeared to be slightly different colours in the past, as the effect would depend on the quality of the water supply! The spectrum below shows peaks in the emission. The solution was intentionally doped with ppm of Ag(I) and Cu(II) ions. The peaks can be attributed to the ion centres trapped in the crystal: at 252, 259 [Ag+] and 359 [Cu+] and 424 nm [(Ag+)2 dimers]. What this tells us is that these ions are trapped in their excited states during rapid transformation from disordered NaCl solute cluster to ordered NaCl crystal. For more details, see the further reading [1].