Laser-induced nucleation
What is nucleation and why is it important?
Nucleation is the process where a new phase of matter is formed from an existing phase: for example, formation of a crystal from solution. Nucleation is of fundamental scientific interest and has significant economic value, e.g., in production of pharmaceuticals and agrochemicals.
Nucleation is the process where a new phase of matter is formed from an existing phase: for example, formation of a crystal from solution. Nucleation is of fundamental scientific interest and has significant economic value, e.g., in production of pharmaceuticals and agrochemicals.
Why is nucleation difficult to study? How can lasers help?
Primary nucleation is difficult to study because it occurs randomly in space and time. Our group use short pulses of laser light to initiate nucleation, in supercooled liquids, supersaturated solutions. In the animated image below you can see crystals being nucleated in real time using a single laser pulse (5 ns). The crystals only form where the beam has passed.
Primary nucleation is difficult to study because it occurs randomly in space and time. Our group use short pulses of laser light to initiate nucleation, in supercooled liquids, supersaturated solutions. In the animated image below you can see crystals being nucleated in real time using a single laser pulse (5 ns). The crystals only form where the beam has passed.
Temporal and spatial control of nucleation
The method we use is called non-photochemical laser-induced nucleation (NPLIN) because no reactions are taking place. Using the method we have demonstrated unprecedented temporal (sub-nanosecond) and spatial (sub-micron) control of nucleation. The image below illustrates the spatial control by nucleation of KCl crystals in an agarose gel [1]. The crystals only form where the laser has passed.
The method we use is called non-photochemical laser-induced nucleation (NPLIN) because no reactions are taking place. Using the method we have demonstrated unprecedented temporal (sub-nanosecond) and spatial (sub-micron) control of nucleation. The image below illustrates the spatial control by nucleation of KCl crystals in an agarose gel [1]. The crystals only form where the laser has passed.
What is the mechanism for non-photochemical laser-induced nucleation?
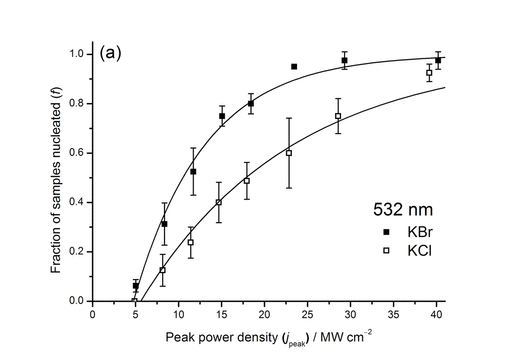
The mechanism originally ascribed by Garetz et al. is based on the optical Kerr effect [2]. It is believed that molecules are present in disordered clusters in solution. The intense transient electric field causes molecules to align: this creates enough order so that the cluster becomes a crystal nucleus and continues to grow. However, this doesn't explain why the effect works on simple salts such as KCl, which have no molecules to align. Another unusual feature is that there is a distinct threshold below which the laser power is not enough to induce nucleation (see figure below).
The mechanism originally ascribed by Garetz et al. is based on the optical Kerr effect [2]. It is believed that molecules are present in disordered clusters in solution. The intense transient electric field causes molecules to align: this creates enough order so that the cluster becomes a crystal nucleus and continues to grow. However, this doesn't explain why the effect works on simple salts such as KCl, which have no molecules to align. Another unusual feature is that there is a distinct threshold below which the laser power is not enough to induce nucleation (see figure below).