Cavity ring-down spectroscopy
What is cavity ring-down spectroscopy?
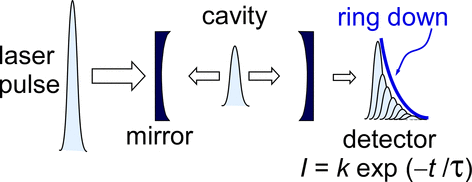
Cavity ring-down spectroscopy (CRDS) can measure very small absorptions of chemical species. It can be many orders of magnitude more sensitive than traditional uv-vis spectrometry. It works by using well-aligned, highly reflective mirrors to recirculate the light in a cavity. Effective pathlengths of kilometers or more are possible. The technique works by measuring the decay (the 'ring-down') of intensity of light in the cavity once you switch the light off. The decay happens partly due to light leaking through the mirrors; they are not 100% perfect. The decay can also be due to absorption or scattering by material in the cavity, i.e., the stuff we are trying to make measurements on.
Cavity ring-down spectroscopy (CRDS) can measure very small absorptions of chemical species. It can be many orders of magnitude more sensitive than traditional uv-vis spectrometry. It works by using well-aligned, highly reflective mirrors to recirculate the light in a cavity. Effective pathlengths of kilometers or more are possible. The technique works by measuring the decay (the 'ring-down') of intensity of light in the cavity once you switch the light off. The decay happens partly due to light leaking through the mirrors; they are not 100% perfect. The decay can also be due to absorption or scattering by material in the cavity, i.e., the stuff we are trying to make measurements on.
What is it useful for?
The main application of CRDS is for atmospheric measurements or for high-precision metrology in the gas phase, e.g., measuring line-strengths of weak (possibly 'forbidden') transitions.
The main application of CRDS is for atmospheric measurements or for high-precision metrology in the gas phase, e.g., measuring line-strengths of weak (possibly 'forbidden') transitions.
Can CRDS be applied to measurements in liquids and solids?
The main issue with trying to use CRDS for condensed phases (liquids and solids) is that there can be large losses due to reflection and scattering of the condensed-phase materials. There have been a number of clever ways that researchers have used to try to circumvent this issue: for an overview see ref. [1]. In general however, you want to keep the volume of sample small, and try to either recycle any reflected light back into the cavity, or minimize the intracavity refelections.
The main issue with trying to use CRDS for condensed phases (liquids and solids) is that there can be large losses due to reflection and scattering of the condensed-phase materials. There have been a number of clever ways that researchers have used to try to circumvent this issue: for an overview see ref. [1]. In general however, you want to keep the volume of sample small, and try to either recycle any reflected light back into the cavity, or minimize the intracavity refelections.
Brewster-angle cavity ringdown spectroscopy
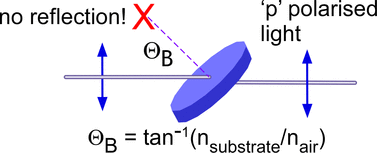
Our group introduced new innovations to enable use CRDS in condensed phases [2-4]. By placing the substrate for analysis Brewster's angle inside the cavity, the surface reflections are minimized (theoretically zero reflection: see image below). We have applied this technique to spectroscopy of thin films with sub-monolayer coverage [2]. We have also measured the reaction kinetics (on nanosecond timescale) of nitrate radicals in solution (at nanomolar concentrations) [3].
Our group introduced new innovations to enable use CRDS in condensed phases [2-4]. By placing the substrate for analysis Brewster's angle inside the cavity, the surface reflections are minimized (theoretically zero reflection: see image below). We have applied this technique to spectroscopy of thin films with sub-monolayer coverage [2]. We have also measured the reaction kinetics (on nanosecond timescale) of nitrate radicals in solution (at nanomolar concentrations) [3].
Flowing liquid-sheet jet
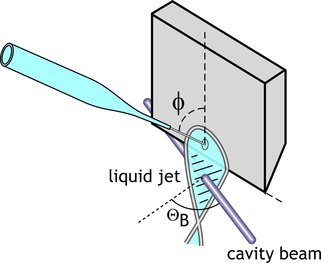
One of our innovations is a thin, flat liquid jet, which is formed by simply spraying a liquid at a flat surface (see diagram below) [4]. The surface tension of the liquid causes the resulting rebound to form an area that is optically flat and approximately 10 microns thick.
One of our innovations is a thin, flat liquid jet, which is formed by simply spraying a liquid at a flat surface (see diagram below) [4]. The surface tension of the liquid causes the resulting rebound to form an area that is optically flat and approximately 10 microns thick.
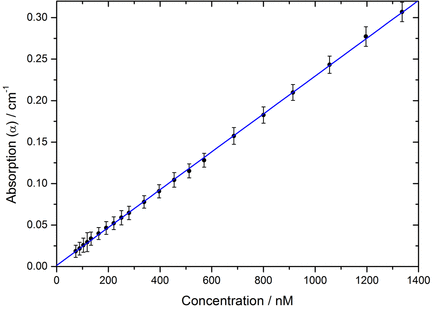
By placing this film at Brewster angle, we can measure concentrations of analytes in the liquid as low as ~70 nM. A particular advantage of CRDS is its high dynamic range: the measurement remains linear at low and high concentrations (see below for solutions of malachite green dye in ethylene glycol). The jet would be useful for study of processes at the vapour-liquid interface.