Bubble nucleation
Laser-induced bubble nucleation
Our group has investigated the nucleation of gas bubbles using pulses of laser light (see pages on NPLIN). Gas nucleation is important in a wide range of applications, including oil and gas recovery, power generation, electrochemistry, polymer production, and waste treatment. From a fundamental physics perspective, the nucleation of bubbles can help us understand the mechanisms of laser-induced nucleation. The animation below shows an example of nucleation in a glass of champagne.
Our group has investigated the nucleation of gas bubbles using pulses of laser light (see pages on NPLIN). Gas nucleation is important in a wide range of applications, including oil and gas recovery, power generation, electrochemistry, polymer production, and waste treatment. From a fundamental physics perspective, the nucleation of bubbles can help us understand the mechanisms of laser-induced nucleation. The animation below shows an example of nucleation in a glass of champagne.
Power dependence
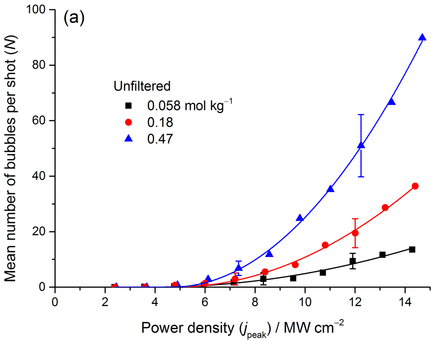
To investigate the phenomenon, we prepared sugary solutions with different concentrations of sucrose. We chilled and carbonated these solutions at high pressure. To determine how many bubbles nucleate we took videos of bubble nucleation using single laser pulses (5 ns) at increasing laser powers [1]. The results are shown below. The laser power is given as peak energy per laser pulse (12 MW/cm2 is approximately 6.3 mJ/cm2). A typical sugary soda has a concentration of 0.32 mol/kg, approximately halfway between the red and blue points. There appears to be a quadratic dependence on laser power; this is different to laser-induced crystal nucleation, which shows a linear dependence, at least at low laser powers.
To investigate the phenomenon, we prepared sugary solutions with different concentrations of sucrose. We chilled and carbonated these solutions at high pressure. To determine how many bubbles nucleate we took videos of bubble nucleation using single laser pulses (5 ns) at increasing laser powers [1]. The results are shown below. The laser power is given as peak energy per laser pulse (12 MW/cm2 is approximately 6.3 mJ/cm2). A typical sugary soda has a concentration of 0.32 mol/kg, approximately halfway between the red and blue points. There appears to be a quadratic dependence on laser power; this is different to laser-induced crystal nucleation, which shows a linear dependence, at least at low laser powers.
Effect of filtration
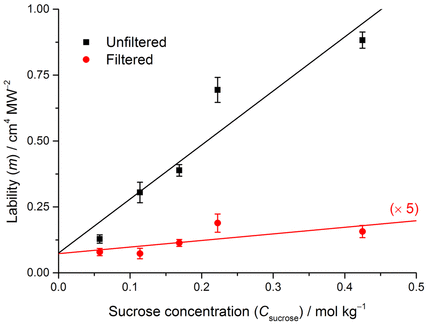
We found that filtering the samples before carbonation gave substantially fewer bubbles, as can be seen in the plot below. The vertical axis is the lability, which is the quadratic coefficient from fitting the data (solid lines shown in plot above). The lability is a measure of how readily a solution will produce bubbles when exposed to a laser pulse.
We found that filtering the samples before carbonation gave substantially fewer bubbles, as can be seen in the plot below. The vertical axis is the lability, which is the quadratic coefficient from fitting the data (solid lines shown in plot above). The lability is a measure of how readily a solution will produce bubbles when exposed to a laser pulse.
Mechanism
NPLIN of crystals has generally been explained by an optical Kerr effect mechanism [2]. This mechanism should not work for bubbles, as pointed out by Knott et al. [3]. The refractive index of the new phase being formed is less dense, which goes against previously held ideas of co-operative effects of polarizability of the solute molecules. We found that the lability appears to be directly proportional to the amount of sucrose, and filtering appears to remove the effect almost entirely. These observations suggest the effect is due to impurity particles in the sucrose. We proposed a new mechanism, based on heating of the nanoparticle impurities.
NPLIN of crystals has generally been explained by an optical Kerr effect mechanism [2]. This mechanism should not work for bubbles, as pointed out by Knott et al. [3]. The refractive index of the new phase being formed is less dense, which goes against previously held ideas of co-operative effects of polarizability of the solute molecules. We found that the lability appears to be directly proportional to the amount of sucrose, and filtering appears to remove the effect almost entirely. These observations suggest the effect is due to impurity particles in the sucrose. We proposed a new mechanism, based on heating of the nanoparticle impurities.
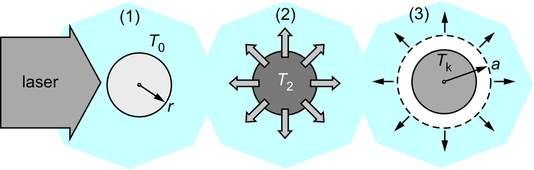
The schematic above illustrates the mechanism: (1) a nanoparticle absorbs energy from the laser pulse; (2) the heat from the nanoparticle is transferred rapidly to a shell of surrounding fluid; (3) the heat transfer causes vaporization of the fluid to create a bubble; this vapor bubble acts as a seed to nucleation of a carbon dioxide bubble. We believe that this mechanism can also explain observations of NPLIN of crystals, and we are continuing to investigate this mechanism.